Patient Data Collection in Clinical Trials (eCOA/ePRO)
Capture patient-reported outcomes digitally, securely, and GCP-compliant.
What is eCOA/ePRO?
Electronic Clinical Outcome Assessments (eCOA) and Electronic Patient-Reported Outcomes (ePRO) enable digital capture of patient assessments during clinical trials. Unlike paper questionnaires, data is captured directly electronically, ensuring higher data quality and compliance with regulatory requirements.
Electronic data collection avoids typical problems of paper questionnaires such as illegible handwriting, incomplete entries, and inconsistent data. Instead, you receive high-quality, timestamped, and traceable data.

High-quality, consistent data

Compliance with regulatory requirements (GCP, GDPR)

Timestamped and traceable entries
The Intuitive eCOA/ePRO Solution of Magana Trial Manager
Two flexible implementation options for your study
eSurvey
One-time surveys of study participants via individual questionnaire links
 Individual link per participant
Individual link per participant One-time response
One-time response Ideal for baseline and follow-up questionnaires
Ideal for baseline and follow-up questionnaires
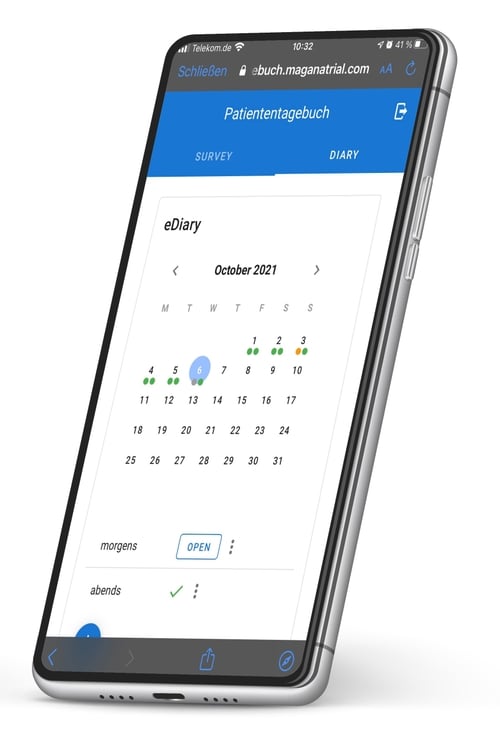
eDiary (Patient Diary)
Recurring surveys of study participants with daily or multiple entries
 Daily or multiple entries possible
Daily or multiple entries possible Automatic reminders
Automatic reminders Ideal for symptom diaries and progress monitoring
Ideal for symptom diaries and progress monitoring
Try it out!
Use the following link or QR code to access a short demo of the eSurvey. Experience for yourself how easy data collection can be for patients.
Scan QR code
eDiary (Patient Diary) for Recurring Surveys of Study Participants
Benefits for Participants

Simple, intuitive operation
User-friendly interface for all age groups

Data entry possible on all devices with internet connection
Smartphone, tablet, or computer

Only questions relevant to each patient are displayed
Personalized questionnaires based on patient profile

Self-monitoring of submitted and missing entries
Overview of all completed and pending questionnaires
Benefits for Study Managers

Quick overview of complete, incomplete, and missing entries
Dashboard with real-time status of all participants

Legible, traceable, and consistent documentation
No problems with illegible handwriting

GCP and GDPR-compliant data collection
Complete audit trail and secure data storage
Your Benefits at a Glance
Usability
- • Study-specific web addresses
- • Access via QR code or link
- • Device-independent data entry
- • Intuitive user interface
Data Consistency
- • Real-time visibility of all entries
- • Easy data exports
- • Complete audit trail
- • Participant-specific controls
Security
- • GCP and GDPR-compliant data collection
- • Encrypted storage on ISO 27001-certified German servers
- • Individual login codes for participants
Experience MaganaMed
Schedule a free demo and learn how Magana Trial Manager can make your clinical trials more efficient.